|
Chandrashekhara Venkata Raman, later FRS and Nobel laureate, was born in 1888 at Thiruchirapalli, in Tamil Nadu. His academic success took an early start when he finished school at the age of eleven and graduated in Physics and English, at the head of his class, at fifteen. Poor health kept him from going to England and he continued at Presidency College, Chennai, to receive his master’s, with honours, at eighteen. Ironically, he now entered the civil service, as Assistant Accountant General, at the age of nineteen with a posting in the Auditor General’s office, at Kolkata |

|
|---|
Happily, for science, he found some spare time to keep in touch with physics and managed to do work on the physics of stringed instruments and Indian drums in the laboratories of the Indian Association for the Cultivation of Science. This institution had been set up by Mahendra Lal Sircar in 1876, with visions that it should become a focus for advancement in science. But it had not progressed at all towards this objective till 1907, when Raman chanced to peep in. Raman saw the IACS sign on his way to work, by tram, one day, and jumped at the possibility of continuing to work with physics. He moved to a house near the IACS premises and would come in early in the morning and stay till he had to leave for work. He would then come after office hours and stay till late in the evening. Thanks to Raman’s work, the IACS attracted an impressive list of men of science and over the years, more than satisfied its founder’s dream.
From 1907 to 1917, C V Raman, worked at the IACS in addition to the duties of his Government job. His work studied a variety of topics but much of the work concerned the science of sound, vibration of strings and drums and optics and the behaviour of light. He published his work in the world’s leading journals and began to be widely known. In 1916, Sir Ashutosh Mukherjee, a brilliant man of letters and long observer of Raman’s work, was appointed Vice Chancellor of Kolkata University. Within a year he managed to create a special chair for the promotion of Physics, the Palit professorship and offered the post to Raman. Raman gladly gave up his promising career in the Civil Service and accepted, for full time devotion to his first love, the pursuit of science.
But getting back to the question about the colour of the sea. The background of the question was that the blue colour of the sky had then been precisely explained by work of Lord Raleigh. Visible light was recognized as consisting of waves of electrical and magnetic disturbances, spreading out rather like the ripples on the surface of a pond, at the tremendous speed of three million kilometers a second. The exact mathematics that applies to waves had perfectly accounted for all of the properties of light, and even its other forms, like radio waves, heat waves, X Rays. These were properties like how it reflects, how it bends in lenses, how radio waves could bounce of the layers of the atmosphere, and so on.
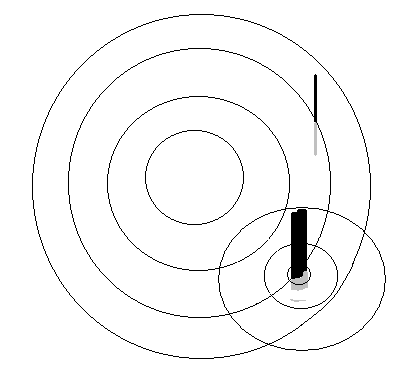
Lord Raleigh, a great consolidator of the classical physics, then looked into how such waves were scattered by specks of dust or even the tiny molecules of the air. To understand scattering, again, we could look at the ripples on the surface of a pond. Consider what happens if there were a twig and a thick post sticking out of the water.
|
The twig hardly seems to affect the passage of the ripples. But in the case of the post, the course of the original wave is broken and another set of ripples spreads out from the post itself. These, secondary ripples, which spread out from the post, are the scattered waves. |
|---|
It seems evident that the post has this effect, but not the twig, mainly because the dimensions of post are comparable to the distance between successive ripples, or their wavelength. The same post, if sticking out of the sea, where the waves are many metres apart, would have no effect, rather like the twig in the case of the ripples!
Raleigh was able to work out that for the same size of obstructions, the strength of scattering increased very fast as the wavelength got shorter. The strength of scattering of a wave with half the wavelength, in fact, was not just twice as much but sixteen times!
The meaning of this discovery, for the sunlight coming through the atmosphere, was that the light towards the violet side of the spectrum was scattered much more strongly than the light towards the red side of the spectrum. The sunlight that finally gets down to surface of the earth is thus a little depleted in components towards the violet side, which explains why the sun looks yellow when we get a quick look at it. Late in the evening, when the sunlight is passing through more of the atmosphere, because of the slant, even more the violet end of the spectrum is depleted and the sun looks orange or red!
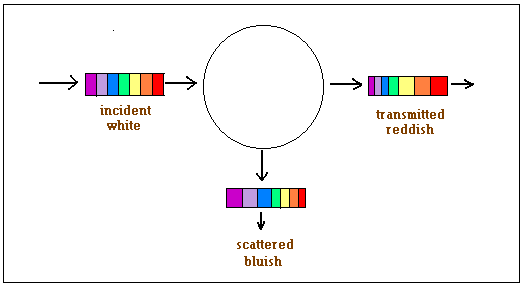
This was the complete, perfect, elegant explanation for the blue sky. But for the blue sea, the current theory simply said that the sea was blue because it reflected the blue sky! When C V Raman saw the dazzling blue of the Mediterranean during his voyage, he thought this glib explanation just did not hold water. Why could it not be a case of light being scattered by the molecules of water, just like the air, thought Raman. And so, while the ship sailed, he made careful observations, using the prism, telescope and spectroscope, which he always carried, and came to important conclusions about the scattering of light by molecules, before the end of the voyage!
The ‘quantum’ theory, which overcame this problem, first said that atoms and molecules did not exist in any of a continuum of energy states, but only in discrete, step-wise, energy levels, separated by ‘quanta’ of energy. And changes in energy levels corresponded to the emission or absorption of the quantum of energy in the form of ‘photon’, or particle of light, of the appropriate energy.
Apart from this ‘need’ for light to have a ‘quantum’ nature, Einstein’s theory of the photoelectric effect, based on light quanta, was a powerful verification. And then, in 1923, Compton had studied an effect where X Rays underwent not just scattering, but a change in wavelength when they bounced off atoms. This effect was so exactly explained by considering X Rays to exist as quanta that gained or lost energy during impact with an electron, just like billiard balls do when they bounce off each other, that there remained little doubt about the validity of the quantum theory. Kramers and Heisenberg, in fact, had also suggested that the same thing was possible in the ordinary scattering of light!
For all this, the classic Raleigh theory was so ‘de rigeur’ and exact, that scientists, Niels Bhor and Max Plank included, were loath to think there could be anything more to say on the subject. It is a mark of Raman’s objectivity and scientific honesty that he felt he must still look for evidence of quantum effects in the scattering of visible light by liquids!
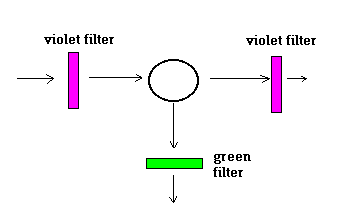
<|
Naturally, some violet light was seen, and even if a violet filter were used, the violet light came through. The objective of the experiments was to introduce a green filter or a yellow filter and see whether scattered light of colours other than violet could be seen. |
|
|---|
The scattered violet light itself was extremely feeble and there was no doubt that if there was any light of other colours to be detected, this would be feeble indeed. The incident beam, derived from bright sunlight, was already the most intense that could be mustered at those times To help the observer detect the feeblest of scattered components, the observer was placed in a darkened chamber, four feet square, for an hour before the experiment was started. This was to prime the observer’s eyes to maximum sensitivity, a method that Lord Rutherford and his students had used in their celebrated discovery of the atomic nucleus! Raman’s box in the IACS laboratory was also facetiously called the ‘black hole of Kolkata’.
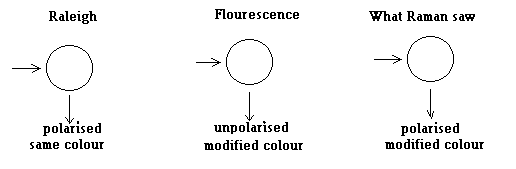
One by one, the researchers began to report that there was a ‘modified’ scattered light, that is, light of a different colour! Classical Raleigh theory was emphatic in saying that the scattered light would be of the same colour. How could the observations be reconciled? A ready explanation was that this was not scattered light but was a case of fluorescence.
Fluorescence is when the atoms in a substance absorb the incident light, and then, usually after a short delay (a very short delay, in microseconds), shift to an intermediate state and emit light at a different wavelength. This is what happens in the common, domestic tube light. The tube is filled with a gas at low pressure, which allows a discharge of electricity, emitting light of a particular colour, often in the ultra violet. This light falls on the fluorescent coating of the tube, which then emits the ‘near white’ light of fluorescent lamps.
Raman and his experimenters could not be sure that the ‘modified’ component they were seeing was really a new wavelength in the scattered light or a case of fluorescence. As fluorescence is often due to impurities, they repeated the experiments with greater and greater purity. But still they saw the ‘modified’ component. By 1925, the team had noticed the effect in fifty liquids.
Raman now began to seriously suspect that they were dealing with a kind of Compton effect in the visible portion of the spectrum. The research work was stepped up and more and more data was accumulated. An important new piece of data was that the ‘modified scattering’ was ‘polarised’ light
|
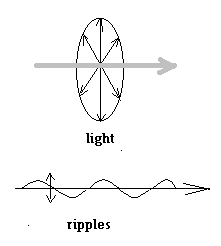
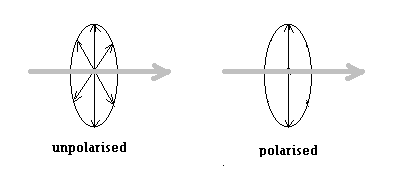
Comparison of light to the ripples on a pond is a pretty accurate description In the case of the ripples, the water at any point is moving up and down, while the ripples move along the surface. In the case of a light wave too, the changes in the electric field are at right angles to, or transverse to the direction of the light beam |
|---|
But unlike in the case of ripples, in the case of light, there are no fixed ‘up’ or ‘down’ directions and the electric wave can be in any direction, so long as it is at right angles to the direction of the beam. In a usual beam of light, we have all possible orientations of the electric wave.
|
But when light is generated not by randomly oriented atoms, but in a given direction only by atoms oriented just so, then, the electric waves in the light beam are all along one axis, and light like this is said to be polarized. |
|---|
Now, Raleigh’s theory of scattering says that scattered light would be polarized and this is what is observed, too. But in the case of fluorescence, the process of absorption and emission of the light wave are separate. Hence, there is no selection of the orientation of the electric wave in the light waves scattered in a particular direction and the emission is unpolarised.
What Raman’s team had seen was that the ‘modified’ ray in the scattering experiments was polarized. This was a strong reason that it was not a case of fluorescence, but really an odd instance of scattering, but with a change in the wavelength!
|
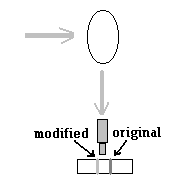
The definitive experiment, of 28th Feb 1928, was with the incident light passed not just through the blue filter, but also through a uranium glass, which made the incident beam not just in the range of violet, but a very narrow band of the spectrum. And when the scattered beam was viewed it was not through a green or other filter, but through a spectroscope, which splits the light coming in into its components, like a prism. And what was seen was, well, one line, which was the original light, and next to it, another, faint line, the new colour generated in the scattering! |
|
|---|
This was a definite, startling experimental result, that in scattering of visible light by liquids, in addition to Raleigh scattering, there arose a scattered component with a change in wavelength, just like in the scattering of X Rays by electrons!
The very next day Raman called a press conference and in weeks the news was the sensation in scientific circles the world over. The explanation, which Raman also published, was easily seen as the incident ‘photon’ having transferred some of its energy to the molecule off which it was scattered, thus leaving the scattered photon with less energy. Using the relation that Plank and Einstein had developed, to connect the wavelength of a photon with its energy, it was straightforward to work out the longer wavelength of the scattered photon with less energy!
And why was there a ‘gap’ between the wavelengths of the incident and the scattered photon? Could the scattered photon not have all energies starting from that of the incident photon? This too was because the molecule that absorbed the energy could do so only in the quanta that separated its ‘energy levels’.
the Raman spectra of materials soon became a sensitive tool to study the low energy, vibration and rotation modes of molecules, an area that thrives nearly unchanged to this day.3>
Recognition was not far in coming, either. Rutherford announced the discovery in the Royal Society, the British Government conferred a knighthood and in 1930, Raman became India’s first Nobel laureate for science! The 1920s were memorable years in the story of physics. Now, for an Indian scientist, working entirely in India, to have a place of honour during those times was electrifying.
Apart from having been an active researcher and having published profusely for two decades, Sir C V Raman had also been the builder of a school of well-trained scientists at the IACS. In 1933, he took over as the first Indian Director of the Tata institute of Sciences, the forerunner of the Indian Institute of Science, Bangalore. During the next fifteen years that Raman spent at the Institute, he did much to set up and develop the Physics Department into one of international repute, apart from training and inspiring a generation of world-class scientists. During these years, he also founded the Indian Academy of Sciences and established the first of the many reputed journals published in the country. He retired from the Institute in 1948 and then set up the Raman Institute a few kilometers away. A notable feature of this institute was that it had been funded entirely by private donations and industry
Raman continued work in research and exposition of science till 1970. On 2nd October 1970, he delivered the customary Mahatma Gandhi Memorial lecture at the Raman Institute. Soon after, he fell ill, and on November 21st, he passed away
------------------------------------------------------------------------------------------