The world’s increasing food demand is creating nitrogen poisoning that competes with global warming, says S.Ananthanarayanan.
Human activity and the use of fossil fuel have created a scepter of melting of polar ice, rising sea level, changing climate and demographic upheaval. But even if alternate fuels were found and warming were reversed, providing food for the rising population could wreck the environment in its own way before the end of the century.
A team at the Potsdam Institute for Climate Impact Research, in Germany and the International Center for Tropical Agriculture, in Colombia, has carried out simulations of the rise in levels of reactive nitrogen, that is, nitrogen in forms other that the inert kind that forms a large part of the atmosphere. This move active form of nitrogen, while necessary for life, can make itself unsustainable when it is in excess. The levels of reactive nitrogen, which is required for agriculture, are already rising much faster than what is safe. The study, which is reported in the journal, Nature Communications, examines where we are likely to land by the year 2050 if we continue in the same way, and alternatively, if we take pollution mitigating measures.
Reactive nitrogen
Nitrogen gas is abundant, it makes up 78% of the atmosphere. And nitrogen is necessary for all known forms of life. It is there in the amino acids, of which proteins are made, it is there in the DNA and RNA, and in plants it is there in chlorophyll. But it is not the nitrogen in the atmosphere that living things can make use of. Gaseous nitrogen consists of pairs of nitrogen atoms bonded as diatomic molecules. The nitrogen atom has three unpaired outer shell electrons, and these pair up when the molecule forms. This triple bond is a very strong bond and it takes energy to get the nitrogen atoms to separate and combine with other chemical groups. The nitrogen in the atmosphere is thus inert, and hardly reacts with anything. This, incidentally, is the reason some foodstuffs are sealed with nitrogen in the wrapping to prevent decay or spoiling.
The chemical bond in the ordinary nitrogen molecule then needs to be broken, to get nitrogen into other compounds, which is to create a form of reactive nitrogen, which is ready to combine and form other molecules, which is important for life processes. This process, which is called nitrogen fixation, happens naturally during thunderstorms, when lightning strikes, and more commonly by action of bacteria in the roots of leguminous plants, which are plants like the pea plant or alfalfa. Agriculture uses up this nitrogen and depletes the soil, which then needs to be left fallow, to regenerate. Or legumes need to be planted in alternate seasons. Or sources of reactive nitrogen need to be added, in the form of manure or chemical fertilizer.
But too much of reactive nitrogen can be a menace too. As the normal nitrogen gas is so stable, or a very low energy condition, the nitrogen in nitrogen compounds is always eager to break free and get back to the happy, diatomic molecule state. Free reactive nitrogen, more than what plants need, thus causes serious damage to health and the ecology. In the air, it raises ozone levels and causes respiratory ailments, even cancer. It can come down as acid rain and damage buildings, acidify the soil or cause lop-sided plant growth, leading to nutrient imbalance and loss of biodiversity. It can render groundwater or surface water unfit for drinking, affecting the oxygen balance in infants, which leads to the blue baby syndrome. When it washes into waterways, it nourishes algae, which feed bacteria, which use up the store of oxygen. And then, nitrous oxide, one of the reactive nitrogen forms, is a greenhouse gas nearly 300 times worse than carbon dioxide!
For centuries, till the industrial revolution, the supply of reactive nitrogen was balanced in the ecosystem and was able to stay within narrow limits, for mankind to flourish. But the last many decades have seen rising population, human prosperity and activity and greatly increased agriculture. For increased agriculture, the natural sources of fixed nitrogen are not sufficient and chemical fertilizer has been made in factories. The Haber-Bosch process revolutionised agriculture by creating ammonia from nitrogen and then huge quantities of ammonia-based fertilizer. As demand for foodgrains was soaring, farmers used fertilizer liberally and costs were even subsidized by the state.
The trouble with the use of chemical fertilizer is that the great part of the chemicals do not stay to be used by the plant, but get washed off by irrigation water, and enter the ground water or waterways. And then there are other sources of reactive nitrogen, in automobile exhaust and the refuse of humans and lifestock! It is estimated that the total flow of reactive nitrogen into the ecosystem is now about 185 million tonnes, as against some 25 million in 1950. And as the world population, and aspirations rise, this figure would also rise and aggravate an already alarming situation.
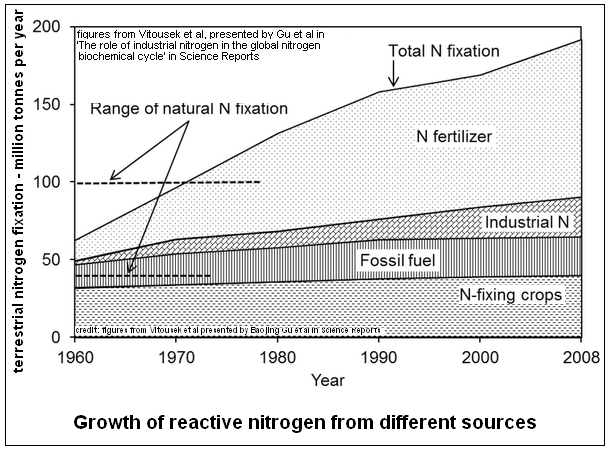
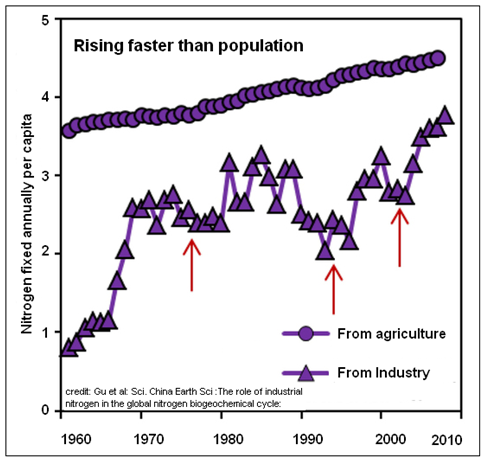
The rise in levels of nitrogen, in fact is seen to be even faster than the population, as the per capita levels also show a rise over the years.
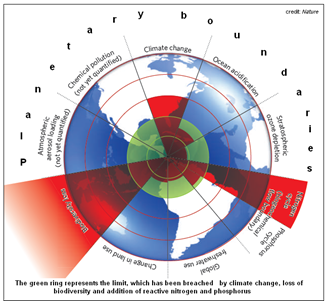
In a paper in the journal, Nature, in 2009, J Rockström and colleagues of the Stockholm Resilience Centre, in Sweden, reported their a study of the earth system processes that were under threat due to human activity and defined the limits that needed to be respected to avoid disastrous and irreversible environmental changes. Of the nine such planetary boundaries identified, three had already been breached, and active nitrogen, where the limit was 35 million tonnes a year, was one. Reviews of the scale of nitrogen lows have since been made and there are outline assessment of future levels, but comprehensive inclusion of available mitigation measures have not been taken into account.
Mitigation
The current study, by the Potsdam and Columbia team, examines the rise in levels till 2050 by modeling land use and the nitrogen budget under different assumptions of consumption patterns and production technology. The result, after considering the estimated population increase, is that nitrogen sources would increase, in the normal course, from 185 million tonnes to 232 million tonnes. The study then considers the mitigating efforts that could be made – (1) Reduced household food waste and recycling of food waste and sewage as fertilizers, (2) lower share of animal-based calories in diets (3) efficient livestock management by improved feeding and higher recycling share for animal manure and (4) efficient fertilization of fields.
These measures are estimated to raise the percentage of nutrient actually used to 69%, against just 22 % at present. This represents a fall in the nutrient that enters the environment and figure for 2050 could come down to 95 million tonnes a year, in place of 232 million tonnes without mitigation. But even this is well above the recommended limit of 35 million tonnes.
While a degree of pollution hence appears inevitable, the paper notes that the impact of pollution depends not only on how much active nitrogen there is, but also where the level is high as also the vulnerable ecology and population. Adaptation to high levels would thus be the way to go, with higher levels being allowed where the risks are lower. And for all that there is need for a global perspective, so that centres of pollution are controlled and do not only shift position, and also to promote mitigation, like reducing food waste, that have far reaching impact.