Kitchen salt may help solar cells get cheaper to build, says S.Ananthanarayanan.
Solar cell technology has seen great strides and the cost has been steadily coming down. Solar cell use has become commonplace, moving from remote locations to rooftops, hand held devices, traffic signals and parking meters. The cost of installation is now down to less than Rs 40/- a watt and solar power is being seen on par with other sources as feeders to the power grid.
Apart from crystalline silicon, there are now other materials that are being used in solar cells. And cadmium telluride, which is one that can match and surpass silicon in the cost per unit electrical output, is set to be the market leader in solar cell materials. But the problem with cadmium telluride (CdTe) is that cadmium is toxic and the process uses cadmium as cadmium chloride (CdCl2), a form of cadmium that can leak into the environment, which calls for expensive precautions. At Euro Science Open Foum 2014, at Copenhagen, J. D. Major, R. E. Treharne, L. J. Phillips and K. Durose, of the Stephenson Institute of Renewable Energy, University of Liverpool said that they described in their paper in the journal, Nature, an alternative to cadmium chloride, which make CdTe solar cells much safer and cheaper to produce.

Solar cells
Solar Cells depend on the ability of some materials to throw out electrons from their atomic structure and create an electric charge, when light falls on them. Now, when there is an arrangement to prevent the electrons so freed from falling back to the atoms that released them, the electrons can be drawn off as an electric current, which is the solar cell. The arrangement to control the movement of electrons is through doping or modifying the nature of the crystals of the photo-material and creating a junction that allows electrons to flow only one way. This enables one side of the junction to collect electrons that get pumped out by the action of light and hence drive an electric current.
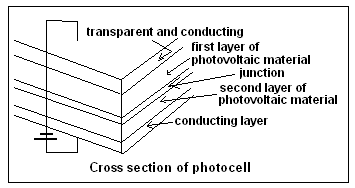
While solar cells were first made of blocks of silicon, they are now used as thin sheets of silicon, or the other materials developed, and the sheets, known as thin film solar cells, can be laid on flexible substrates. The sheets can then be used on roofs of building, small devices or even as a film on window panes, which cuts the glare and also generates electricity.
In the use of CdTe as the material, the junction is with cadmium sulphide (CdS). The efficiency of the junction is greatly increased in a process known as activation with the use of cadmium chloride, which allows chlorine and oxygen atoms to take part in the mechanism of the junction. Although cadmium is toxic, the forms of CdTe and CdS are stable and it has been shown that when used in thin film photovoltaic cells, as in sheets laid on rooftops, the materials do not escape the environment even if there is a fire. But this is not true of CdCl2, which is used in the manufacturing process. CdCl2 powder is water soluble and a positive hazard, both for the workers in the industry as well as to the environment. The control and retrieval of CdCl2 is thus a major element of cost in the manufacture of the CdTe solar cell. CdCl2 is also an expensive raw material, in any case.
But the use of CdCl2 in annealing the junction has made for raising its efficiency of converting solar energy to power from less than 2% to more than 10%, which makes it commercial. Laboratory trials have even touched 20% and there is every interest in cutting cost and risk in the use of CdCl2. The only alternative that was tried is a chlorofluorocarbon gas, which is related to CFC, the material of aerosols and refrigerants that leads to damage to the ozone layer. As use of CFCs is internationally controlled, this alternative of CdCl2 is not available and CdCl2 is still there in the commercial high efficiency CdTe solar cell programme.
New kid
The Liverpool team experimented with other low cost chlorides as possible replacements for CdCl2. The chlorides tried were of magnesium, sodium, potassium and manganese. Others, like chlorides of copper or zinc either have environment issues or high cost. Of the chlorides tried out, it was found that magnesium chloride, MgCl2, gave results almost the same as with CdCl2. Magnesium was found to be suitable, because even with CdCl2, there is magnesium that enters through the soda-lime glass substrate, and magnesium does not affect performance. This is found to be because of the magnesium ion being electrically inactive in CdTe, unlike ions of sodium, potassium, or manganese, whose use led to device performance being compromised.
In the case of magnesium chloride, the efficiency in fact, was 13.5%, compared to only 13.2% even with CdCl2, the traditional activation agent. The long term stability was also found to be the same, the degradation over six months being only that due to oxidation at the gold-CdTe contacts that are used. Magnesium chloride is also a low cost and commonly available salt. As some news reporters observe, it is an ingredient salt that is used in the bean curd, TOFU.
The discovery of the Liverpool team is thus that magnesium chloride can immediately replace cadmium chloride in the manufacture process and eliminate both the risk and the costly toxin control measures involved. The stage is hence set for development for industrial use and wider deployment of CdTe devices.
One factor that affected CdTe was the scarcity and limited reserves of Tellurium and this factor was considered as a serious limitation. But new reserves have been discovered in China, Mexico and Sweden. Interestingly, astrophysicists have also found that Tellurium is the most abundant of the heavier elements in the universe! Nearer home, rich reserves, more than we will ever need, have been found in undersea ridges and feasibility of recovery is being looked into, as other possible reserves. CdTe technology, now easier with MgCl2, appears to have found a place in the drive to increase the use of solar power.
Do respond to : response@simplescience.in
------------------------------------------