Bio-degradables are stepping in to rack up solar cells, says S.Ananthanarayanan.
The crashing prices of solar panels held out the promise of affordable solar farms and, in tandem with growing wind power, of viable power generation sans pollution. However, although mass manufacture has brought down the cost of solar cells, their energy conversion efficiency is low, not more than 15%, in forms practically available. The use of solar cells is hence still not commercially economical. This apart, fabrication of solar cells itself is power intensive, and again, the solar cell material, at the end of the cells’ life, is also a pollutant.
In the context, the development of a device that is found to double the output of solar cells, and which is also simple to fabricate as well as to dispose of, is doubly significant. Jingwen Ding, Jie He and Challa V Kumar, at the University of Connecticut, report and have just presented at the 250th National Meeting & Exposition of the American Chemical Society (ACS). ACS, the world's largest scientific society, their success in testing an easily assembled film that helps solar cells get mileage out of previously inaccessible portions in the spectrum of sunlight.

The reason the sun gives out heat and light is that it is fiercely hot. Hot things radiate energy because the atoms or other electrically charged components of which things are made up are in rapid vibration. The hotter they are, the more rapid the motion and the more they radiate. It has been observed and also worked out that while the photons, or particles of light, of which the radiation is composed, are there at almost all wavelengths, there is central wavelength, which represents a given energy of photons, at which an object at a given temperature would radiate the most. And as the temperature is raised, the maximum radiation moves to higher energy, which is to say, lower wavelength.
Thus, objects that are only warm, like a cup of tea, radiate in the long wavelength, infrared region. A red-hot object radiates in the shorter wavelength of red light, while a white hot object radiates in even shorter wavelengths. The sun is at about 6,000°C, and the maximum radiation is in the region of yellow light, with almost as much in the other rainbow colours, and also energy in the shorter wavelength, ultraviolet, and the longer wavelength, infrared.
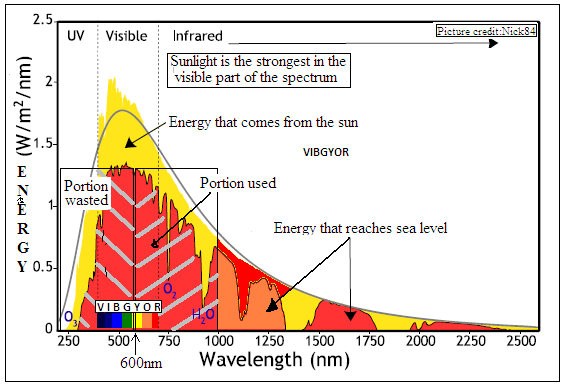
Living things, like plants have adapted to use this region, from violet to red, for photosynthesis and also for vision, so that they make the best use of the sun’s radiation. And now, that we are trying to tap the same radiation to power our high energy lifestyle, it is fortunate that the available materials to build photo-voltaic devices with also respond to nearly the same part of the spectrum
Solar Cells
When photons of light of the appropriate energy strike certain metals, they surrender their energy to the metal by knocking one of the electrons in the atoms of the metal, so that electrons pile up at surface of the metal. Now, if the pile of electrons is drained by a conductor leading from the surface to the body of the metal, this is an electric current, which can heat things, drive machinery, charge batteries, etc. It is based on this principle that the photo-cell and hence the solar cell panels have been developed. The most suitable materials, dimensions and design have been worked out and the last decade has seen rapid improvement.
The limitation, however, is that metals have not evolved, like living things, to make the best use of sunlight, and the suitable range of wavelengths for silicon which is the most common metal used in solar cells, is from 1,000 to 600 nanometers. The radiation of shorter wavelength, 600 to 350 nm, which is also intense, is wasted. What is more, the radiation in this blue end of the spectrum and also in the ultraviolet is the more energetic and has the effect of causing damage to carefully constructed photo-voltaic cell material. There is hence interest, both in making use of the energy in short wavelength radiation as well as in protecting photo cell material.
Green antenna
What the Connecticut researchers have done is to make use of a property, somewhat like one called fluorescence, of certain substances to absorb light at one wavelength and then emit part of the energy absorbed at a longer wavelength. Normal fluorescence is well known in the domestic fluorescent tube, where discharge of electricity through the gas in the tube gives off ultraviolet light. The tube itself is coated, in the inner surface, with fluorescent materials which absorb the UV light and emit light at wavelengths that approximate to white light.
Professor Kumar and team have made use of a related phenomenon in certain materials, where the lower energy emission is not in the form of a lower energy photon, but a transfer, without any radiation, to a neighbouring atom, and then radiation at an appropriate wavelength by the second atom. This phenomenon, which is called Förster Resonance Energy Transfer, is an effect seen only at very small dimensions, where the two systems are in coherence, and it requires the twin conditions of the two systems being close enough together and still not in contact, when FRET cannot take place.
Building on earlier work, reported in the journal of the Royal Society of Chemistry, where Dr Kumar was a participant, the Connecticut team has identified a set of dye molecules which are able to act as ‘donor’ and ‘acceptor’ in FRET, as a cascade from light of wavelength at the blue end of the spectrum to emission at wavelengths at the red end. Work like this has been done in the past, but the medium has been in solution, and the participating dye molecules were often allowed to come in contact, inhibiting FRET. They have also been expensive to produce. The Connecticut team has employed a simple method of mixing a protein, albumin, derived from animal serum and fat from the common coconut, to create a gel that sets as a film when warmed. Proteins have complex molecular structure that allows other molecules to attach to specific portions of their exterior, which is why specific proteins are able to in specialized ways in living things. In this case, the team believes that the dye molecules are similarly trapped by the protein-fatty acid matrix and kept closely packed, but still held apart, so that FRET action can take place. And as the material sets as a film, it is in solid form and can be easily draped over a solar cell, to filter the lower wavelength incident light and deliver it to the solar cell at the proper, longer wavelength.
“We can absorb solar light from 350 nm or blue region to 600 nm or the near red region. The photons absorbed in this wide color window are then converted to red photons of about 600 nm,” says a note from the research team. The fabrication of the film is a very simple process of mixing the components and then warming at some 80°C for twenty minutes, when the film forms. "It can be done in the kitchen or in a remote village. That makes it inexpensive to produce," Dr Kumar says.
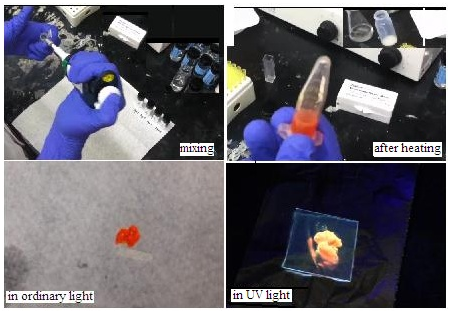
The transparent film becomes a light pink or reddish when mixed with dyes, because of emission on the red side of the spectrum or yellow-orange, when seen in UV light. The effect can be destroyed if any one of dyes is left out, which shows that they all take part in the cascade. Testing the film on actual solar cells has shown positive increase in electrical output and the estimate is that there can be doubling of the output of common solar cell types. And the doubling of output comes with the addition of natural materials that degrade without environmental damage – “they are even edible”, Dr Kumar says.
------------------------------------------------------------------------------------------