The elusive timekeeper hidden in an atomic nucleus has been traced, says S.Ananthanarayanan.
Science and modern life make exacting demands on accurate timekeeping. The current standard, which is based on the exact separation between atomic energy levels, is accurate to over one part in many billions. The energy levels, however, are affected by environmental factors. If there were a similar energy difference within the atomic nucleus, which is comparatively better shielded than the atom, it could be possible to have a steadier category of clocks.
Lars von der Wense, Benedict Seiferle, Mustapha Laatiaoui, Jürgen B. Neumayr, Hans-Jörg Maier, Hans-Friedrich Wirth, Christoph Mokry, Jörg Runke, Klaus Eberhardt, Christoph E. Düllmann, Norbert G. Trautmann and Peter G. Thirolf, from institutes in Munich, Darmstadt and Mainz, Germany, report in the journal, Nature, that they have pinned down a transition of the thorium nucleus that gives off a photon of light in the ultra violet region. Such a transition, which could be the basis of a nuclear clock, has been expected, based on theory, and was being sought for since over a decade, but could not be realized in practice.
What is needed for time keeping is something that has an unchanging rhythm, so that the number of times that it beats is a measure of time that has elapsed. The first of these, of course, was the length of the day, or the time from sunrise to sunrise, which was seen as unchanging. For convenience, this length of time was divided into 24 hours and then, with invention of devices that could measure smaller intervals, into minutes and seconds. The earliest devices were the regular flow of water, sundials and hourglasses. The pendulum was an important development and with ingenious gears and movements, clock making grew in sophistication and became an art form.
The first real breakthrough was with electronics and the quartz crystal vibrator, which provided a rock steady beat of several thousand or even million strokes a second. The crystal was linked to an electronic oscillator, and the crystal controlled the frequency of the oscillator to be in step with its own. Crystal clocks, which were developed in the 1930s, soon found wide application. Quartz clocks became the standard for international time and also of the frequency of radio and TV transmissions and enabled progress in science and engineering. Great refinement of astronomical observations became possible, variations in the length of the day, for instance, could be detected over as short a period as a few weeks, in place of the months of observation needed earlier.
The step that followed the crystal clock was the atomic clock, which uses the frequency of the microwave radiation from atoms to regulate the working of an electronic circuit that generates microwaves. The caesium-133 (133Cs) atom, the stable form of the element, caesium, has 55 orbiting electrons, which have a pair of energy levels whose difference corresponds to a frequency of light of 9,192,631,770 cycles a second. This radiation has wavelength of about 3.26 cm, which is in the range of microwaves and hence matches the capability of electronic oscillators. This fact, that the caesium atom can absorb radiation at a frequency that suits available technology, is the main reason that caesium time has come to be the standard.
In the caesium clock, caesium vapour is passed through an intense microwave field in a chamber which has high vacuum. Before entering the microwave field, the caesium atoms pass through a magnetic field that selects the atoms in the base state of the energy gap that we are interested in. The microwave field is tuned to very near the correct frequency and sweeps through a narrow band of frequencies, so that it is periodically at exactly the correct frequency. When the frequency matches, caesium atoms absorb energy and transit to the higher energy state. At the exit of the microwave chamber, the atom beam is again filtered to measure the number of atoms in the higher state.
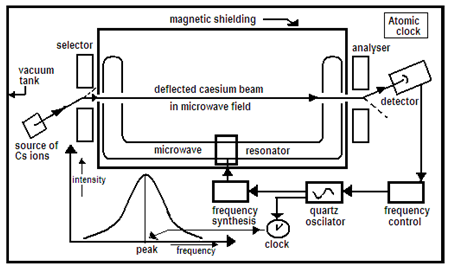
The peaking of the number in the higher state is used to tune the oscillator, till the frequency is correct and the change of state of Cs atoms stays at the peak. The frequency is then exactly 9,192,631,770 cycles a second and the oscillator locks. The frequency is then divided, by the number of cycles, for instance, to give an accurate one beat per second.
Atomic clocks are now accurate to about a billionth of a second in the course of a day, which works out to one part in 10,000 billion ( one in 1014). International Atomic Time (TAI, from the French name Temps Atomique International), is the average of the time kept by over 400 atomic clocks worldwide. This standard is the basis of the Coordinated Universal Time, known as UTC (a compromise with Temps Universel Coordonné, in French), which, in turn, is used for civil timekeeping, like aviation and flight control, for the Internet, and also for Terrestrial Time, which is the standard for astronomy and cosmology.
Nuclear option
For all their accuracy, however, the energy levels in atomic clocks are affected by electric fields, or the radiation from the surroundings, even if the caesium vapour or other gas is cooled to cryogenic temperatures, to contain the motion of the sample atoms. An option is to use the same design of the clock, but to use energy levels in atomic nuclei, which are shielded from outside influence, in place of the energy levels of the electron shells of the atom.
This is acceptable in principle, but the problem is that the binding of the particles in the nucleus is much stronger than of electrons outside the nucleus and the differences in energy levels in the nucleus are generally thousands, even millions of times greater than in electron shells. Transitions would thus correspond to gamma ray photons, or at best, X ray photons. As we have no oscillators, or even lasers, at these frequencies, the use of nuclear energy levels for time keeping has appeared to be out of reach. Except that, in theory, the thorium-229 (229Th) nucleus had a pair of energy levels whose difference was that of a photon in the ultra violet. The higher energy state was ‘metastable’ or sufficiently long lived for the transition to the state to be detected and the wavelength, 150 to 170 nm (violet light is 400 nm), was not outside the reach of a laser to be developed.
Since the time that this transition was proposed, in 1976, and despite progress made in 1994 and 2007, however, the existence of this excited state of the thorium nucleus was only inferred and not directly detected, the authors of the paper say. But the possibility “can potentially bridge the fields of nuclear and atomic physics, as it conceptually allows for optical laser excitation of a nuclear transition. This in turn has stimulated thoughts about transferring existing knowledge of laser manipulation of the electronic shell to a nuclear system, leading to interesting proposed applications such as a nuclear laser, nuclear quantum optics, and a nuclear clock,” the authors say in the paper.
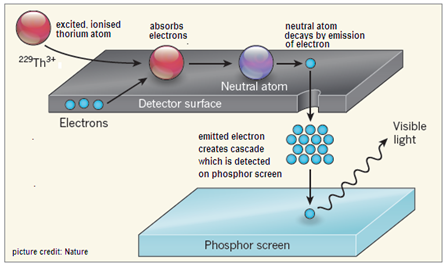
The authors carried out an experiment with 229Th atoms generated from the radioactive decay of Uranium-233 (233U). The thorium atoms arising from this decay are in an ionized state, where the atom has lost two or three of its orbital electrons. While the nucleus is also in an energetically excited state, the loss of electrons does not allow the ionized atom to lose energy by the ejection of another electron. The decay may hence result in a radioactive change in the nucleus. The neutral, 229Th atom, on the other hand, is not constrained and would readily de-excite by causing the ejection of an electron.
The experimental arrangement hence provided, apart from the filtering out of other products of uranium decay, for the neutralization of the thorium ions and then watched for the decay of excited, neutral thorium atoms by an arrangement to capture the electron thrown out. The experiment provided ample safeguards to eliminate other products of the decays and to ensure that the electrons detected were the result of the higher, slightly longer lived energy state of 229Th. “……we were then able to measure the decay of the excited isomer back to the ground state of 229Th as a clear and unambiguous signal,” says co-author Peter G Thirolf from Ludwig-Maximilians-Universität München.
There is still much work to be done, to harness this nuclear transition, which corresponds to a frequency in the ultra violet. Many other properties of the decay need to be studied to implement a process of creating resonance for use as a nuclear clock. The first step, however, to show clearly that the transition itself exists, has been taken.
Why do we need a faster clock?
The 229Th nuclear clock would be orders of magnitude more accurate than the best atomic clocks. Considering that caesium clocks are already accurate to one part in 1014 and clocks based on other atoms and also clocks on optical frequencies are even more accurate, is it worth the effort to work on nuclear clocks? A comment on the paper, by Marianna Safronova from the University of Delaware, carried in Nature, says, “every time that clock precision has improved, new and frequently unexpected applications have emerged.”
This apart, an ever present role for more accurate clocks is to probe for variations of fundamental constants, like the charge of the electron or Planck’s constant, which are possibilities to resolve some conundrums in physics. Ultra-precise clocks can also be useful in the quest for ‘dark matter’, which appears to account for the bulk of the mass of the universe
Nearer home, more accurate time keeping would refine the GlobalPositioning System, a technology based on signals exchanged by twenty four satellites in orbit around the earth. Another important use is for survey and prospecting. As the gravitational force of masses affects the pace of time, the most accurate clocks would be able to create 3D maps of an area by a simple aerial survey, and even detect underground deposits of ores, or oilfields.------------------------------------------------------------------------------------------