There may be a whole different form of chemistry on the surface or within the largest stars, says S.Ananthanarayanan.
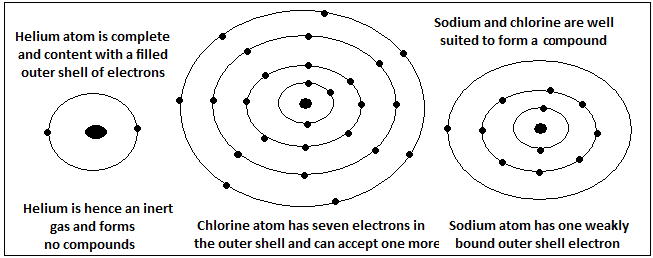
Hydrogen is the simplest of elements, with just one proton contributing to its weight. Hydrogen gas is hence the lightest and often used to launch balloons. Helium is the second in the hierarchy of elements, with four heavy particles in its nucleus. Helium is also a light gas and is also used to fill lighter-than-air balloons.
A problem with hydrogen gas in balloons is that hydrogen is highly inflammable. This problem is amply avoided if one uses helium, as helium does not burn at all. In fact, in the matter of being safe for balloons, helium goes to the other extreme. Helium, with neon, is the most inert of all elements and is not known to form any stable compound.
Intense search in the last few decades has resulted in a few evanescent instances, the most successful one being a compound with a positively charged hydrogen ion. This compound too, most readily decomposes, aggressively pushing away its loosely bound hydrogen ion to neutralise other elements or compounds that can use the hydrogen ion. In theory, it is possible to conceive of compounds with lithium, caesium, fluorine, but the calculated energy of these compounds are high and the compounds must needs be only instantaneously stable.
At least, all this is so in ordinary conditions. Short lived compounds have been known to form under high pressure and there has been the expectation that a stable compound could arise under very high pressure. An international team of researchers in different institutes in Russia, China, Italy, Germany and USA, led by Prof Artem R. Oganov of the Skolkovo Institute in Moscow, describe in the journal, Nature Chemistry, their quest for such compounds and success in finding one.
The principle in the combination of elements is that lower energy conditions are more stable. If the total energy of a pair of atoms together is less than the energy when they are separate, they will combine, rather than stay apart. Atoms are a group of positive charges, held together by short-range, nuclear forces, with an equal number of negative charges, the electrons, in orbit. Depending on the number of electrons, the atom is generally not in the lowest energy condition that is possible. It is found that electron shells are best packed in groups of two or eight. When an atom has more electrons than can be packed like this, electrons spill over into incomplete, outer shells. The tendency to achieve the numbers of two or eight then drives a tendency of the atoms to combine with others atoms, which have a complementary excess or shortfall, with respect to the numbers of two or eight
Helium, with two positive charges in the nucleus, has just two electrons in the first and only orbit, and this is a lowest and very stable condition. This is to say that it takes a lot of work to pull an electron away from a helium atom and also that the helium atom is such a low energy entity that combining helium with another atom would take some work, rather than giving off some energy that is saved as a result of the marriage.
Change under pressure
When atoms are under heavy pressure, however, there has been some work done in squeezing them together and the economics of combination can change. Under pressure of almost 2 million times the atmospheric pressure, for instance, the heavier of the ‘noble’ gases, so called because they are inert, become reactive, and xenon forms oxides, and krypton, xenon and argon combine with magnesium. The metal sodium has a single, loosely bound electron in its outer orbit and is hence a good conductor of electricity. But under heavy pressure, sodium becomes an insulator. With only one electron available for combination, the sodium atoms (Na) form a stable salt, sodium chloride, denoted as NaCl, or one sodium atom in combination with one chlorine atom (Cl). Combinations of different numbers of sodium atoms with more chlorine atoms are not possible. But this changes under pressure, and unusual and stable combinations like, Na3Cl, Na2Cl, Na3Cl2, NaCl3, and NaCl7 are formed when the pressure is raised!
The group of researchers made use of an algorithm called USPEX (for Universal Structure Predictor….), developed by Prof Oganov, which is able to work out the energy of formation and hence stability of structures that could be possible, given the characteristics of constituents. The group carried out assays of helium combining with different elements, hydrogen, oxygen, fluorine, sodium, potassium, magnesium, lithium, rubidium, caesium, etc. The results were that it is only sodium that forms a stable compound with helium at pressures which were practical.
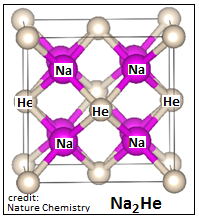
Calculations showed that a combination of two atoms of sodium with one of helium, Na2He, had a total energy that was less than the energy of a mixture of sodium and helium, or any other, at a pressure of about 1.6 million atmospheres. And also that once formed, the compound would remain stable down to a million atmospheres.Diamond anvil
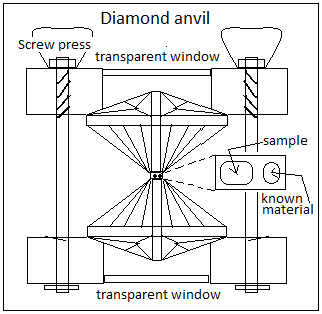
The researchers loaded sodium in a medium of helium in the confines of a ‘diamond anvil’ and subjected the sample to pressures in millions of atmospheres. The anvil consists of two opposing bits of diamond, between whose tips a sample of test material can be place and compressed. The hardness and transparency of diamond ensures that the pressure is transmitted and also allows the sample to be studied with the help of light or X rays. A material whose behaviour under pressure is known is placed in the anvil along with the sample, and this material enables the pressure applied to be measured. The diamond also allows lasers to be shone on the material for heating.
X rays consist of light waves whose dimensions are comparable to the distances between atoms in solids and the structure of materials can be deduced from the way X rays are scattered by the material. The Raman effect is that vibration of atoms brings about changes in the wavelength of scattered light. The paper in Nature Chemistry describes the changes in the visual, X ray and Raman scattering as the pressure was increased – from showing that the sodium compound was formed and to the structure and properties of the crystals formed.
Our current belief, that it is in ‘earth-like’ conditions that life can arise, is rooted in the state of materials being manageable and organic chemical processes being possible only in the narrow temperature range that we have on the earth. While helium does not participate in processes that take place on the earth, it now appears that helium could be chemically active in places, as on giant stars, where mega pressures are encountered. As helium is the second-most abundant element (after hydrogen) in the universe, this throws up the possibility that some radically different form of sentient entities may exist in distant worlds!
------------------------------------------------------------------------------------------