Viruses can be the vehicle to carry genetic material to the inner ear and remedy hearing loss, says S.Ananthanarayanan.
Inherited genetic deficiency is the reason for about half the 250 million cases of hearing loss, worldwide. In most of these, the function itself of the organs of the ear is affected and medical intervention is generally not effective. Even devices like hearing aids or implants in the middle and inner ear which partially restore auditory function, are not always possible.
Answers to many medical conditions, even haemophilia, leukaemia, Parkinson’s disease have been found in gene therapy, where genetic material is introduced into living cells, to modify their function. In dealing with hearing loss, however, there have been limitations in the use of gene therapy, because of the different organs involved, particularly the inner and outer hair cells, which act in concert to ensure the remarkable sensitivity of the ears of mammals.
Two groups pf researchers, working in institutes in Massachusetts, Chicago, New Orleans, North Carolina, Washington and Vienna, report in the journal, Nature Biotechnology, success in creating in the laboratory a virus that is able to invade the cells of the inner ear and introduce a scrap of genetic material that sets right the working of both kinds of deficient hair cells. The work has been done with a particular inner ear condition that affects mice, but this first time that hearing function has been recovered holds promise of being useful to deal with human hearing loss, the authors of the papers say.

While the architecture of the ear provides the framework for capturing and channelling sound waves, it is the fine hair-like structures in the inner ear that translate sound vibrations into nerve signals, to be recognised as sounds. And here, there are different kinds of hair cells, some to sense high and low frequencies and others to help focus different frequencies at specific places. The complexity is incredible – the tiny, liquid-filled resonating chamber of the ear, the cochlea, has tens of thousands of hair cells and different frequencies of sound are focussed at points just hundredths of millimetres apart.
The functioning of physiological systems is orchestrated by the action of proteins, which play different roles of signalling and enabling organs to act in specific ways. Thousands of proteins are synthesised within the cells by joining together chemical units, called amino acids, according to specific patterns. The patterns are spelt out by the units in sequence along the length of the DNA, the giant molecules that code the entire genetic information of the cell. If there is an error or an omission in the code for a protein which is important for the cell’s function, then the cell is not able to generate that protein and is not able to work correctly.
It is this kind of deficiency that is found to bring about deafness in some 50% of the hearing impaired people in the world. And more than 300 locations along the length of the DNA have been found to be relevant and over a hundred genes, or bits of DNA that code for the production of particular proteins, have been isolated as involved in people affected by loss of hearing.
Gene therapy seeks to remedy this condition by replacing the deficient gene with a functional, therapeutic version, so that that a genetically deficient cell creates the required proteins and functions normally. Therapeutic genes, once introduced into the cell, are guided by additional bits of DNA-like sequences to seek out the correct place and replace the deficient portion. A difficult part of the operation, however, is the actual insertion of the genes and bits of DNA-like material into the diseased cell.
The vector
One successful method of doing this is with the help of viruses. Viruses are ‘almost’ living entities, that resemble cells, in so far as they contain DNA, but not any further. Viruses are almost only DNA, contained inside a protein coat, sometimes with a fatty covering. They have no other apparatus to create proteins, or even to reproduce. But the special feature is that they are small, most cannot be seen with an optical microscope, and they are able to get through the outer membrane and into the body of specific kinds of cells. It is within such a ‘host’ cell that viruses use the resources of the cell to reproduce, and this is the process, which could lead to failure or death of the cell, that causes the disease arising from viral infections.
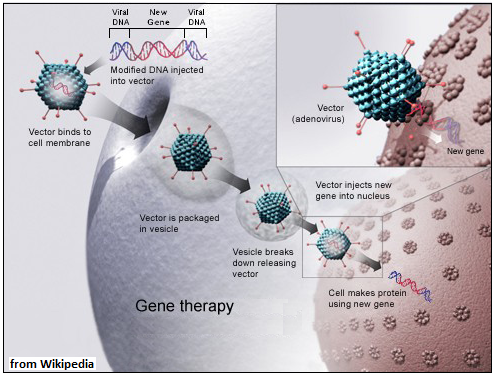
This disease causing capability can also be used for the therapeutic purpose of inserting genes into gene deficient cells. The viruses used are a category called adeno-associated viruses, which have been found to be present with other viruses, but cause no disease. AAVs are small, just 20 nanometres and are able to enter many kinds of cells. And then, they attach to the DNA not at random, but at a specific place, known as AAVS1. This quality makes AAVs safe and convenient to use for gene delivery.
One group of the researches, Lukas D Landegger, Konstantina M Stankovic and Luk H Vandenberghe and others, tried out a number of AAVs and found that synthetic virus, Anc80L65, was able to efficiently transfer a fluorescent green protein into both inner as well as outer hair cells. Being effective with outer hair cells was an improvement over existing vectors. Lab tests were carried out with human cells and the vector was found to be equally effective.
The second group, Gwenaëlle S Géléoc and colleagues, sought out a genetic condition where the genes involved were found in both types of cochlear hair cells. The Usher’s syndrome, which causes severe deafness and also affects balance and leads to blindness, results from the lack of a gene, UCH1C, which encodes the protein, harmonin. A subgroup of gene deficient mice, the c.216AA, were found to show both the hearing and visual defects of human Usher’s syndrome. Study of c.216AA mice showed that both types of cochlear hair of these mice lost their function after the first week of birth.
As lack of harmonin was the apparent cause, the group investigated whether introducing harmonin in c.216AA hair cells would preserve their function. The known, Anc80L65, synthetic viral vector was hence designed to carry the code for harmonin-A1 or harmonin-B1, and introduced into the inner ear by injection.
The results were dramatic, thousand-fold improvement of hearing, to near normal levels, of otherwise deaf and dizzy c.216AA mice. More than a hundred genes may be implicated in disorders of the human ear. Finding ways to use Anc80L65 with large animals could speed up the discovery of gene therapy methods for disorders of the human ear, the authors of the paper say.
Complexity of hearing
The human ear, which is not the most sensitive, among animals, can react to sounds where the pressure difference is less than one billionth of the atmospheric pressure. Such a faint sound corresponds to movement of air by the distance of a tenth of an atomic diameter! And then, the arrangement is incredibly sturdy, the sounds can get so loud as corresponding, for an instant, to 10,000 times the atmospheric pressure. This sensitivity, in the human ear, is over the range of frequency, or pitch, from 20 cps to 20,000 cps. Dogs can hear up to 40,000 to 60,000cps, while cats go as high as79,000 cps and dolphins and bats, which use high pitched sound for navigation, can hear sounds at 100,000 cps. Mice also make and hear sounds as shrill as 79,000 cps, for communication, outside the range of normal predators. (The sensitivity of cats is probably an adaptation to get the mice anyway).
But range of frequency apart, the remarkable feature of the ears is the ability to tell the difference of very small change in frequency. The spiral of the cochlea is only about 3.2 cms long, but it is able to separate about 1,500 different frequencies, using 16,000 to 20,000 hair cells. This amounts to a separate frequency being focused every 0.002cm. Even with just a dozen or so hair cells assigned to each frequency, such high resolution would need some form of sharpening of the response to pitch along the length of the cochlea, a mechanism that is still not understood. S.A.
------------------------------------------------------------------------------------------