A rare form of water ice, so far imagined in theory, has been realized in the laboratory, says S.Ananthanarayanan.
Water, abundant on the earth and basic to all life, displays great polyvalence in the forms that it can take. It is present as vapour in the atmosphere, as a liquid in rivers and oceans and as a solid, in the form of ice. This last form, as a solid, is known to have eighteen different compositions.
While ice remains essentially the same, H2O, in all these forms, at very high pressures, there is the possibility of the hydrogen and oxygen components of water separating. Such forms are imagined to be possible in the interior of the giant planets and there have been theoretical studies of what the properties of such ice should be. Marius Millot, Sebastien Hamel, J. Ryan Rygg, Peter M. Celliers, Gilbert W. Collins, Federica Coppari, Dayne E. Fratanduono, Raymond Jeanloz, Damian C. Swift and Jon H. Eggert, at the Lawrence Livermore laboratory, California, the University of California at Berkley and the University of Rochester report in the journal, Nature Physics, that they have used laser driven shock compression to create instances of ice in the form conjectured at very high pressures
The most striking and commonly known feature of water is the way its components arrange themselves as liquid water approaches the temperature of freezing. Water reduces in volume, like most other materials, with lowering temperature and energy, till it reaches 4°Centigrade. From that temperature onwards, however, and till it freezes, water begins to expand, or grow less dense. And after it freezes, at 0°Centigrade, it shows a series of crystalline forms as it cools to lower temperatures.
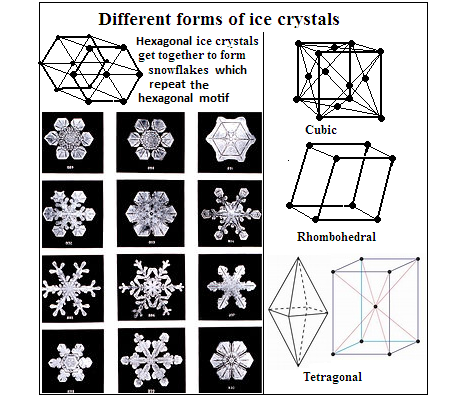
The normal ice, with which we are familiar, exists in hexagonal crystals, which can arrange themselves in kaleidoscopic ways as snowflakes. The next form of ice is in cubic crystals arranged in diamonds, which forms below minus 53° C and changes to normal ice when warmed to minus 33°C. The next form of ice has a regular rhombohedral structure and forms when normal ice is cooled below minis 83° C and compressed.
Then there are forms of ice that get made with compression at high pressure and these forms are denser than water. When moderately cooled and compressed to about 3,000 atmospheres, ice forms as tetragonal crystals, the lightest of the high pressure forms.
There is also a kind of ice that is non-crystalline, or amorphous. This exists in three forms, the low density, high density and very high density. When high density amorphous ice is warmed at a pressure of over 8,000 atmospheres and there are particles, like dust, it takes a rhombohedral crystal form. At just minus 20 C and 5,000 atmospheres, we get the structure of a rectangular prism. At just minus 3 C and 10 million atmospheres, we get a tetragonal structure.
And in this way, water ice takes on different crystal forms at different temperatures and pressures. In all these forms, however, the electrical forces between the positively charged hydrogen, H+, and negatively charged, O- components of water molecules are able to keep the parts of water together and the behavior is non-conducting, like normal water. In extreme conditions, of millions of atmospheres, however, displacements of the components of the water molecule can be so energetic that the electrical forces are overcome and H+ and O- ions float apart. This is a form known as ‘ionic water’ and has been so far a theoretical conjecture of a form that should exist. It is conjectured that at even higher pressure, ‘superionic water’ would form, where the oxygen crystallises and charged hydrogen float about in a lattice of oxygen atoms.
As this form of water would be electrically conducting, the reality in the interior of Neptune and Uranus, the giant ice planets should be truly unusual. It is hence important to test the milestones of the theoretical understanding, to devise ways to verify conjectures about the superionic state, or the dynamics of the interiors of the giant planets. It is conjectured that superionic water would be as hard as iron and glowing yellow in colour.
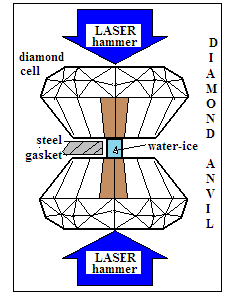
The paper in Nature physics outlines the work done so far in compressing water to high pressures in diamond anvils, but says that unambiguous verification of the high electrical conductivity expected in the superionic state has not been possible. Even dynamic, of shock compression has not been able to bring water to the stable superionic state as the rise in pressure has been too fast. In the present study, the paper says, laser driven shock compression was distributed over a series of steps, resulting in measuring much higher electrical conductivity. The unit used for measuring conductivity is the Siemens and the practical unit for water is the micro-S or the milli-S. The conductivity measured in the shock compression trials came to ~ 30 S/cm and in the reverberating shock trials it came to ~150 S/cm.
The emergence of high conductivity alone was not enough to show that the superionic state had been reached, as high conductivity was also possible with conduction by electrons in place of conduction by positively charge hydrogen atoms, or protons, the paper says. Further study was hence carried out, to observe the conditions in which electronic conduction set it and these were found not to be there in the reverberating shock experiments. The indication was hence that “Altogether, the new data provide experimental evidence for superionic conduction in water-ice at planetary interior conditions,” the paper says.“Considering the abundance of H and O in the Solar System, and the stability of H2O molecules, water is expected to be an important planetary building block,” the paper says. “As astronomical observations and extreme-condition laboratory experiments improve in the near future, we are confident that tying planetary modelling with the fundamental properties of planetary constituents should soon yield a better understanding of the diversity of icy planet formation, structure and evolution.” The paper says.
------------------------------------------------------------------------------------------ Do respond to : response@simplescience.in